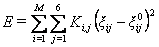Clicking on the service name, a new window will be automatically opened, showing the complete web service description, input object/s, output object/s, secondary parameters (type, units, default, etc.) and general information.
Set-up for Simulation |
Check for disulphide bonds
WS: addDisulphideBondsFromPDBText
Checking for Disulphide Bonds with a distance criteria.
- Distance Cutoff: 2.5 Å
- SS bonds added to the PDB data as a REMARK.
|
Clean PDB
WS: cleanStructureFromPDBText
- Remove Crystal Waters.
- Remove Crystal Hydrogens.
- Remove Non-Parametrized Ligands.
|
Fix Side Chains
WS: fixSideChainsFromPDBText
Fix Side Chain Problems, adding missing heavy atoms (if possible).
Program: Leap (Ambertools). |
Add structural water molecules and ions
WS: titrateStructureFromPDBText
Add structural water molecules and ions in the energetically most favourable positions at the surface of the structure.
Program: CMIP, Classical Molecular Interaction Potentials.
- By default, necessary ions (Positive ions: Na+. Negative Ions: Cl- ) are added in order to neutralize the system.
|
Set ionization state
Set the appropriate ionization states according to pH
Programs: ProtPka (Fast). CMIP (Slow) |
|
Only Histidine residues
WS: protonateHistidinesFromPDBText |
|
All ionizable residues
WS: protonateIonizableResiduesFromPDBText |
Energy Minimization (Hydrogen Atoms)
Energy minimization of hydrogen atoms keeping all heavy atoms fixed to their original positions |
| |
NAMD
WS: optimizeStructureFromNAMD_MD_Structure
Usable for both AMBER and CHARMM topologies
Program: namd2 from NAMD Package.
- Available types of minimization: Conjugate Gradients.
|
| |
GROMACS
WS: optimizeStructureFromGROMACS_MD_Structure
Programs: grompp and mdrun from Gromacs Package.
- Available types of minimization: Conjugate Gradient and Steepest Descent.
|
Energy Minimization (All atoms)
Possibility of restraining atom movements with a given force constant. |
| |
NAMD
WS: optimizeStructureFromNAMD_MD_Structure
Usable for both AMBER and CHARMM topologies
Programs: namd2 from NAMD Package.
- Type of minimization: Conjugate Gradients.
|
| |
GROMACS
WS: optimizeStructureFromGROMACS_MD_Structure
Programs: grompp and mdrun from Gromacs Package.
- Types of minimization: Conjugate Gradient and Steepest Descent.
|
Solvate Structure
Adds a solvent shell to the system according to the specific settings. By default, the necessary ions (Positive ions: Na+, Negative Ions: Cl-) are added in order to neutralize the system |
| |
AMBER
WS: solvateStructureFromAMBER_MD_Structure
Structure Solvation in Amber.
Program: Leap (Ambertools)
- Available box types:: Shell, Spheric, Cubic and Truncated Octahedron.
- Box size specified by Å of spacing distance around the molecule.
- Water molecule type: TIP3P
|
| |
NAMD
WS: solvateStructureFromNAMD_MD_Structure
Program: VMD (solvate and autoionize plugins) from NAMD Package.
- Available box types: Cubic, and Spheric.
- Box size specified by Å of spacing distance around the molecule.
- Water molecule type: TIP3P.
|
| |
GROMACS
WS: solvateStructureFromGROMACS_MD_Structure
Programs: genbox and genion from Gromacs Package.
- Available box types: Cubic, Triclinic, Dodecahedron and Truncated Octahedron.
- Box size specified by nanometers of spacing distance around the molecule.
- Water molecule type: SPC.
|
Molecular Dynamics simulations
Simple Box Solvent System Equilibration (NPT, NVT, or NVE)
Simple Box Solvent Molecular Dynamics (NPT, NVT, or NVE)
- Particle Mesh Ewald (PME) for full-system periodic electrostatics.
- Available Ensembles: NPT, NVT, NVE
|
| |
NAMD - Amber
WS: runMDFromAMBER_MD_Structure
Program: namd2 from NAMD package.
ForceField: Amber Parm99SB* for Proteins, Amber Parm99bsc0 for Nucleic Acids (DNA/RNA)
- Constant temperature dynamics via Langevin Dynamics.
- Constant pressure dynamics via Nose-Hoover Langevin piston.
- SHAKE is used to maintain all bonds involving hydrogen atoms at their equilibrium value
|
| |
NAMD - Charm
WS: runMDFromNAMD_MD_Structure
Program: namd2 from NAMD package.
Forcefield: Charmm
- Constant temperature dynamics via Langevin Dynamics.
- Constant pressure dynamics via Nose-Hoover Langevin piston.
- SHAKE is used to maintain all bonds involving hydrogen atoms at their equilibrium values
|
| |
GROMACS
WS: runMDFromGROMACS_MD_Structure
Program: mdrun from Gromacs package.
- Constant temperature dynamics via Velocity-rescale algorithm.
- Constant pressure dynamics via Parrinello-Rahman algorithm.
- LINCS Linear Constraint Solver was used to maintain all bonds at their equilibrium values.
|
Coarse Grained Molecular Dynamics Simulations |
Coarse-Grained MD: Brownian Dynamics (C-Alpha)
WS: runBrownianMDFromPDBText |
Protein is put in a stochastic bath that keeps the temperature constant and modulates the otherwise extreme oscillations of the residues. This bath is simulated with two terms accounting for a velocity-dependent friction and stochastic forces due to the solvent environment. The potential energy used to compute forces assumes a coarse-grained representation of the protein (Calpha-only) and a quasi-harmonic representation of the interactions.
- Time: total simulation time.
- Delta_t: Time ellapsed between two consecutive time steps.
- Output Frequency: Number of frames after which a frame is written and considered for analysis.
- Force Constant: Measure of the strength of the spring connecting atoms measured in Kcal/mol*Ų.
|
Coarse-grained MD: DISCRETE dynamics (C-Alpha)
WS: runDiscreteMDFromPDBText |
Protein is modelled as a system of beads (Calpha atoms) interacting through a discontinuous potential (square wells). Outside the discontinuities, potentials are considered constant, thereby implying a ballistic regime for the particles (constant potential, constant velocity) in all conditions, except at such time as when the particles reach a potential discontinuity (event or collision). At this time, the velocities of the colliding particles are modified by imposing conservation of the linear momentum, angular momentum, and total energy. DMD has a major advantage over techniques like MD because, as it does not require the integration of the equations of motion at fixed time steps, the calculation progresses from event to event.
- Time: total simulation time.
- Delta_t: Time ellapsed between two consecutive time steps.
- Cut-off Distance: Maximum distance for the pairs of atoms to be included in the calculation.
- Sigma is the well amplitude for consecutive Calpha atoms.
- Sigma Go is the well amplitude for non-consecutive Calpha.
|
Coarse-grained MD: DISCRETE dynamics (Heavy atoms)
WS: runDiscreteMD_AA_FromPDBText |
Protein is modelled as a system of beads (ALL Heavy Atoms) interacting through a discontinuous potential (square wells). Outside the discontinuities, potentials are considered constant, thereby implying a ballistic regime for the particles (constant potential, constant velocity) in all conditions, except at such time as when the particles reach a potential discontinuity (event or collision). At this time, the velocities of the colliding particles are modified by imposing conservation of the linear momentum, angular momentum, and total energy. DMD has a major advantage over techniques like MD because, as it does not require the integration of the equations of motion at fixed time steps, the calculation progresses from event to event.
- Time: total simulation time.
|
Coarse-grained MD: NMA (C-Alpha)
WS: runNormalModeAnalysisFromPDBText |
Normal Mode Analysis (NMA) can be defined as the multidimensional treatment of coupled oscillators from the analysis of force-derivatives in equilibrium conformations. This methodology assumes that the displacement of an atom from its equilibrium position is small and that the potential energy in the vicinity of the equilibrium position can be approximated as a sum of terms that are quadratic in the atomic displacements. Vectorial Anisotropic Network Model using coarse-grained (Calpha) protein representation is the formalism implemented here. Through the diagonalization of the hessian matrix, the ANM provides eigenvalues and eigenvectors that not only describe the frequencies and shapes of the normal modes, but also their directions.
Possibility to choose between three ANM formalism definitions of the force constants:
- 1. Linear algorithm uses a unique potential is used for all interactions considered.
- 2. Kovacs algorithm uses a distance-dependent potential.
- 3. Mixed algorithm uses a potentials adjusted to atomistic MD simulations.
- Force Constant: Measure of the strength of the spring connecting atoms measured in Kcal/mol*Ų.
- Cutoff: Maximum distance for the pairs of atoms to be included in the calculation.
|
Montecarlo DNA Dynamics (DNA)
|
A mesoscopic Metropolis Montecarlo algorithm associated to DNA helical parameters can be used to obtain coarse-grained DNA dynamics. In this algorithm, the geometry of each DNA base pair is defined by 3 local rotations (roll, tilt and twist) and 3 local translations (slide, shift and rise) and the conformational energy is estimated from the deformation matrix using an harmonic model:
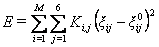
where index "i" stands for one of the M base pair steps and index "j" for the six unique helical parameters (ξ) for each step. The equilibrium values for one helical parameter in a given base pair step type and and the associated deformation constant (Kij) were previously determined from molecular dynamics simulations. To optimize the simulation, maximum amplitudes for Montecarlo movements are selected as to obtain roughly a 40% acceptance rate. Once a movement in helical coordinates is accepted by Metropolis test, the corresponding Cartesian structure of the DNA structure is generated using an adaptation of X3DNA.
- Number of Snapshots: Total number of snapshots in the final trajectory.
|
Molecular Dynamics Trajectory Simple Analysis |
| |
Plot RMSd along the trajectory
WS: getTrajectoryRmsFromMD_Trajectory
Root Mean Square deviation along the trajectory.
Programs: Ptraj from AmberTools package (Binpos,Crd,DCD,NetCDF trajectory formats). g_rms from Gromacs package (XTC trajectory format).
- Possibility to select a subset of atoms/residues from the whole system.
- User defined Mask only available for ptraj-compatible trajectory formats (Binpos,Crd,DCD,NetCDF).
|
| |
Plot RMSd x Residue
WS: getTrajectoryRmsPerResidueFromMD_Trajectory
Average Root Mean Square deviation per residue along the trajectory.
Programs: Ptraj from AmberTools package (Binpos,Crd,DCD,NetCDF trajectory formats). g_rmsf from Gromacs package (XTC trajectory format).
- Possibility of use a reference structure (PDB format).
- Possibility to select a subset of atoms/residues from the whole system.
- User defined Mask only available for ptraj-compatible trajectory formats (Binpos,Crd,DCD,NetCDF).
|
| |
Plot BFactor per residue
WS: getTrajectoryBfactorPerResidueFromMD_Trajectory
Gets Bfactor values per residue from a Trajectory.
Programs: Ptraj from AmberTools package (Binpos,Crd,DCD,NetCDF trajectory formats). g_rmsf from Gromacs package (XTC trajectory format).
- Possibility to select a subset of atoms/residues from the whole system.
- User defined Mask only available for ptraj-compatible trajectory formats (Binpos,Crd,DCD,NetCDF).
|
| |
Plot Radius of Gyration along the trajectory
WS: getTrajectoryRadiusOfGyrationFromMD_Trajectory
Programs: Ptraj from AmberTools package (Binpos,Crd,DCD,NetCDF trajectory formats). g_gyrate from Gromacs package (XTC trajectory format).
- Possibility to select a subset of atoms/residues from the whole system.
- User defined Mask only available for ptraj-compatible trajectory formats (Binpos,Crd,DCD,NetCDF).
|
Trajectory format conversions |
| |
Compress trajectory to PCZ
WS: zipTrajectory
Program: pcazip. Pcazip (https://mmb.irbbarcelona.org//software/pcasuite) compresses Molecular Dynamics (MD) trajectories using Principal Component Analysis (PCA) algorithms. Pcazip offers a good compression ratio at the expense of losing some precision in the trajectory.
- Quality param: Specifies the quality of the compression as a percentage of explained variance (default 90%)
- Gaussian RMSd param: weighted RMSd, giving less weight to atoms in flexible regions (e.g. loops).
- Number of Eigen Vectors Param: Specifies the number of eigenvectors that must be stored in the file. With 0 value pcazip will get the necessary number of vectors to obtain the percentage of explained variance (quality param).
|
| |
Decompress PCZ trajectory
WS: unzipTrajectory
Uncompress trajectory from PCZ format to CRD format.
Program: pcazip (https://mmb.irbbarcelona.org//software/pcasuite). |
| |
Convert trajectory to a set of PDB Files
WS: fromMD_TrajectoryToPDB_Collection
Program: ptraj from Ambertools package. |
| |
Convert trajectory to BINPOS Format
WS: fromMD_TrajectoryToMD_TrajectoryBINPOS
Program: ptraj from Ambertools package. BINPOS is the Scripps Binary format. |
| |
Converts trajectory to CRD Format
WS: fromMD_TrajectoryToMD_TrajectoryCRD
Program: ptraj from Ambertools package. CRD: Amber ASCII format. |
| |
Converts trajectory to DCD Format
WS: fromMD_TrajectoryToMD_TrajectoryDCD
Program: ptraj from Ambertools package.
DCD: CHARMM/X-PLOR/NAMD Binary format. |
| |
Converts trajectory to NetCDF Format
WS: fromMD_TrajectoryToMD_TrajectoryNetCDF
Program: ptraj from Ambertools package.
NetCDF (Network Common Data Form) is a machine-independent binary data format for array-oriented scientific data. |
Flexibility Analysis |
| |
Flexibility Analysis
WebServer: FlexServ
- Principal Components.
- Variance Profile.
- B-factors Landscape.
- Lindemann Coefficients.
- Apparent Stiffness.
- Hinge Point Prediction.
- Residue Correlations.
- Collectivity Indexes.
|
Trajectory manipulation
Programs: Ptraj from AmberTools package (Binpos,Crd,DCD,NetCDF trajectory formats). g_rmsf from Gromacs package (XTC trajectory format).
- Possibility to select a subset of atoms/residues from the whole system.
- User defined Mask only available for ptraj-compatible trajectory formats (Binpos,Crd,DCD,NetCDF)
|
| |
Get Average Structure
WS: getAverageStructureFromMD_Trajectory |
| |
Get a trajectory fragment
WS: getSliceFromMD_Trajectory
Gets a set of snapshots from a Trajectory. |
| |
Get a trajectory snapshot
WS: getSnapshotFromMD_Trajectory |
| |
Remove Water molecules and ions from trajectory
WS: getDryTrajectoryFromMD_Trajectory |
| |
Return trajectory for a set of atoms
WS: getMaskTrajectoryFromMD_Trajectory |
 MDWeb is powered by a set of BioMoby Molecular Dynamics Web-Services (MDMoby). In this help section a short description of the web services available in MDWeb is shown.
MDWeb is powered by a set of BioMoby Molecular Dynamics Web-Services (MDMoby). In this help section a short description of the web services available in MDWeb is shown.